Diffraction of Electrons by a Crystal
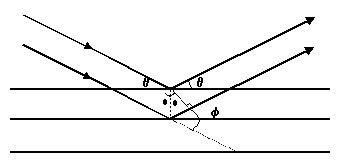
| atomic layers = |
|
| electron wave packets diffracted from atomic lattice |
|
|
| double the energy (E) of the electrons |
|
|
|
 |
 |
(1.) electron particles behave like waves
(2.) can measure (A) wavelength or
(B) inner atomic spacing