Statistics of Particles...
Classification by Spin
Pauli Exclusion Principle: no two
particles in the same state (identical spin, color
charge, angular momentum, etc.) can
exist in the same place at the same time.

The nucleus of an atom is a fermion or boson:
- whether the sum of the number of protons and neutrons is
odd or even
- explains the strange behavior of very cold helium
- a superfluid (no viscosity) because nuclei are bosons and
pass through each other.
Fermoins...The Matter Particles:
- Characteristics:
- subject to Pauli exclusion
- cannot co-exist in the same location
- any particle that has odd-half-integer (1/2, 3/2,
...) intrinsic spin
- measured in quantized units of
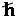 ;
;
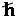 = 1.055 x 10-34 J s
= 1.055 x 10-34 J s
- spin is the internal angular momentum of a
particle in units of
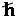 .
.
- Examples of fundamental matter particles:
- quarks and leptons
- most composite particles
- protons and neutrons are fermions.
Bosons...The Fine Particles
- Characteristics:
- have an integer spin measured in the units of
h-bar (spin = 0, 1, 2...)
- not subject to Pauli exclusion
- Examples of fine particle matter:
- The carrier particles related to all the
fundamental interactions
- Composite particles with even numbers of fermion
constituents (such as mesons)



