Textbook Chapters 10 & 11
Chapter 10. The Nucleus of the Atom: Scattering Reveals the Microworld
As Bohr, De Broglie, Schroedinger and Planck were discovering the quantum laws that described the allowed states and wave functions the negative electrons confined in the atom, others were exploring the positive charge that rendered the overall atom electrically neutral. At the turn of the century, scientists conjectured that atoms were permeable spheres with a uniform distribution of electrons and positive charge within. In 1909 Rutherford attempted to verify this hypothesis. His famous scattering experiment, and its surprising result, became the paradigm for a century of more powerful microscopes and their revelation of unimaginable new particles and forces.
Rutherford's Discovery of the Nucleus
Using new found, highly energetic projectiles (alpha particles) from a radioactive source, Rutherford collimated a stream of them and shot them toward a tissue-paper thin foil of gold, as is shown in Fig. 10-1. After passing through the layer of gold atoms, the ionizing alpha particles (charged +2) impinge upon a zinc sulfide screen. Where hit, the screen glows from the de excitation of the atoms ionized by the alpha particles.
Fig. 10-1. A schematic representation of Rutherford's famed apparatus that revealed the nucleus.
Like all scientists at the time, Rutherford expected that the beam of energetic (megavolt) alpha particles would zip unimpeded through the gold foil, and that the beam would only light up the small region of the screen in the beam line, as in the left side of Fig. 10-2. As Rutherford watched, most of the dots of light from alphas were in the path of the beam. But to his and everyone's surprise, some dots revealed scattering at large angles, even backwards! It was as if the alpha projectiles were light, ping pong balls, generally passing through nothing, but occasionally they were scattered by heavy bowling balls buried at large distances from each other in the gold foil. Rutherford's analysis of his results, the right side of Fig. 10-2, led to the revelation that a small, dense, positively-charged nucleus exists at the middle of each atom. This new hypothesis, that atoms have nuclei, is a keystone in the development of modern atomic theory.
Rutherford's technique of inspecting a target (and thereby "seeing" it) by scattering projectiles from it is precisely how we all perceive our world. For example, when we "see" a tennis ball on the court, photons from the sun travel from it (the source) to the tennis ball (the target), bounce off of it, and hit your eye (the detector), revealing the presence of the tennis ball. The scattering of different wavelengths of light from the source encode the color of the ball. By reconstructing the directions and wavelengths of the photons, you eyes "see" that tennis ball.
Nature uses this scattering technique for sound waves, as well as for light photons: Dolphins and bats emit and detect sound waves to "see" their world with acoustic "eyes" as their sonar waves are reflected from obstructions and food in their environment.
Rutherford Scattering: Reinvented with Each New Technology
Using waves to explore the physical world has its limitations. Image resolution can be no better than the wavelength of the source. Our eyes are genetically attuned to the sun's visible light, with wavelengths ~ 500 nm, too wide to analyze anything
smaller than a cell. For higher magnification, must use waves with smaller wavelengths, e.g. the matter waves of an electron microscope to see crystals and viruses. But even best microscope only shows only a fuzzy picture of the atom, as we have seen in the scanning tunneling microscope picture of Fig. 8-4.
In Chapter 6 we discussed water waves diffracting around a large object, leaving a shadow behind. By contrast, meter long waves in a swimming pool easily pass by a stick in the water, unaffected by such a tiny target. Similarly, light waves, with their limited wavelength, are of marginal use to probe the nucleus. Hence Rutherford and his successor employ De Broglie's matter waves, whose wavelength is unbounded on the small side, since ? = hc/E. (see p. 8-1). Moral: If you want small wavelength to make a better microscope, use matter particles and give them a large energy.
Over the last century scientists have done just this. Since they cannot use light to explore sub-atomic structures, they have turned to higher and higher energy particles as a necessity to resolve smaller substructures.
Unstable Nuclei: Radioactivity
Initially, energetic particles (like the alpha particles used by Rutherford) came from nature itself. Late in the 1800's Roentgen discovered a new type of light radiation (X-rays he called them) that could penetrate a black box. These rays were emitted when an energetic electron beam struck the glass at the end of the vacuum bottle in which they were accelerated. Boiled off by a hot electric filament, and accelerated by +1000 volts from the filament to the end of the evacuated tube, the instantaneously stopping electrons were an antenna that radiated high energy photons. (The X-ray tube was much like that of electron diffraction apparatus used in one of your laboratory experiments.)
Radiation similar to X-rays, but with much higher penetration power, mysteriously also comes from rocks! Becquerel covered different minerals with opaque wrap and placed them on photographic plates. To his amazement, something penetrated the black coating and exposed the photographic plates if the rocks contained uranium, radium, and thorium. Energetic rays, without any energy input, were somehow being produced. He concluded that some elements were inherently unstable, and spontaneously released energy.
Three Types of Radioactive Particles
Marie and Pierre Curie discovered that three distinct types of accelerated particles are produced in radioactive decay, naming them after the first three letters of the Greek alphabet: ??(alpha), ??(beta), and ? (gamma). They are distinguished by their electric charge and by their penetrating power. In a magnetic field, the +2 charged alpha particle (a helium nucleus, with 2 protons and 2 neutrons) bends one direction, the negatively charged beta particle (an electron) bends the opposite direction, and a neutral gamma ray (a high energy X-ray photon) does not bend at all, as is shown in Fig. 10-3. (Photons with increasing energy, and decreasing wavelength, differ in name only: radio waves, infrared, visible light, UV, X-rays, and gamma .)
Fig. 10-3. The bending by a magnet of the three types of particles observed from radioactive decay by the Curies.
The Curies' three types of radiation are also distinguished by their penetrating power. As shown by experiments in class with a Geiger counter and everyday sources, alpha particles from a smoke alarm are stopped by a sheet of paper, beta particles from a gas lantern mantle are stopped by the glass window, but even a cm of lead cannot stop the gamma radiation from the Uranium glaze on a 1950's Fiesta bowl. Gamma's are so penetrating they can go far into a material, disrupting the chemical bonds by ionization. A fourth type of nuclear radiation, the neutron (an uncharged, slightly heavier version of the proton), was discovered later. It, like alpha particles, can be boiled off by a fissioning nucleus. Sadly, many years passed before the Curies and their fellow scientists realized the perils of penetrating radiation.
Each species of spontaneously decaying nucleus is characterized by a half-life, as the radioactivity decreases exponentially with time. The half-life measures how long it takes for half of the decaying nuclei to disappear. When a particular atom will decay is an utterly random process. Why will an atom, just sitting there, decay according to some set probability? This is quantum mechanics (Chapter 8) at work. At the time, many physicists were very unhappy that chance rules physical properties. Einstein proclaimed, "God doesn't play dice!" But Einstein was wrong.
Various nuclei with different half-lives provide convenient clocks by which we measure the times of prehistoric events. For example, dating with radiocarbon 14C, the nucleus with 6 protons and 8 neutrons (too many) spans the 10,000 year period. (The number to the upper left of an atomic symbol is the sum of the number of protons and neutrons, i.e. the nucleon number.) With uranium 238U, whose lifetime is 4.5 billion years, we date the solar system! 238U decay initiates a chain of disintegrations that includes the noxious Radon 86 gas along the way. The decay chain eventually stops at the ultra stable lead 206Pb, a "doubly magic" nucleus. One of several successful models of the nucleus, analogous to that of the electrons in the atom, hold that the nucleus has sequentially filled shells, occupied with either protons or neutrons. The nucleus of Lead 206 contains both a full shell of protons as well as a closed shell of neutrons.
Alpha Decay: Quantum Mechanical Tunneling
Most naturally occurring nuclei are stable. However, the
bigger a nucleus, the farther its protons are apart. Eventually,
the repulsive force the the + charges wins out over the short-range attractive
"strong" force that holds the nucleons together. This is especially
the case if the nucleus has too few or too many neutrons.
A particularly efficient way of eliminating excess neutrons and protons
is to shoot out a tightly-bound alpha particle.
In another successful model of the nucleus, the protons and neutrons bounce around inside the nucleus like balls in an MIT "bag", with a minuscule probability that any nucleon is ever outside the bag. Nonetheless, 2 protons and 2 neutrons could, by quantum mechanical chance, appear together outside the nucleus; the probability is greater for a very large nucleus than a small one. Once outside the short range of the strong force, the +2 alpha particle is forcefully repelled by the remaining positive nucleus. A heavy nucleus, may, for the shortest of instants, exist in a state that allows it to blow apart. This, nature's accelerator, was ably used by Lord Rutherford.
The nucleus is held together by the strong interaction; it would blow apart by electrical repulsion between protons if it were not "glued" together by gluon particles that carry the strong force. By analogy, each nucleus is a tightly cocked spring (the electrical repulsion) held in place by very stiff rope (the strong force). The energy stored-up in the spring is released when an alpha particle is emitted.
(As we will discuss in the next chapter, each proton or neutron is not fundamental, but is a composite, on average, of three, point-like quarks, again observed by a high-energy Rutherford-type experiment. The quarks in each proton or neutron are held together by the carriers of the strong force, the gluons.)
E = mc2 : From Mass Springs Energy
Many heavy elements...uranium, thorium, radium ...decay into simpler elements spontaneously. When this happens, tremendous energy is released (MeV...millions of electron volts... as opposed to the single volts characteristic of the chemical energy of an electron confined to an atom). From where does this energy come? The mass of a uranium nucleus = 238.0508 atomic mass units (amu). It typically decays into an alpha particle (4.0026 amu) and a thorium (234.0436 amu) nucleus. Uranium's mass, minus the mass of the two decay products, = 0.0046 amu. Where did this "missing" mass go? Einstein said: a lot of energy is stored in the strong force holding the uranium nucleus together. When the U nucleus decays, this binding energy, potential energy stored as mass, is released as kinetic energy (the energy of motion of the two decay products).
Another classical image of the nucleus, similar to the bag picture, is the liquid drop model. In Fig. 10-4 two uranium-235 nuclei are shown absorbing a neutron, becoming unstable, and splitting in two roughly equal fragments. On average, three neutrons are also liberated, each of which can then stimulate another fission. A chain reaction is initiated, the heat from which can transform water to steam to drive a turbine and electric generator, or, alternatively, do something more disastrous as a nuclear weapon.
Fig. 10-4. Two representations of radioactive fission of a heavy
nucleus envisaged as a liquid drop. The lower representation shows
the decay cascades of the two fission fragments.
In the lower graphic of Fig. 10-4, two of the subsequent radioactive
decay cascades are depicted. Each fragment decays sequentially until
a stable daughter nucleus remains. Stability is reached when energy
is no longer released in the decay process, i.e. the nuclear reaction is
no longer exothermic. As is shown in Fig. 10-5, a schematic version
of the periodic chart of the nuclei, stability is characterized by a diagonal
"valley" in a table plotting the number of neutrons in a nucleus horizontally
(roughly proportional to the mass of the nucleus) and the number of protons
vertically (the electric charge). For low numbers of protons,
a nucleus is stable with equal numbers of neutrons. As a nucleus
gets bigger, one can energetically stuff more than this equal share of
neutrons between the protons, since neutrons are electrically neutral.
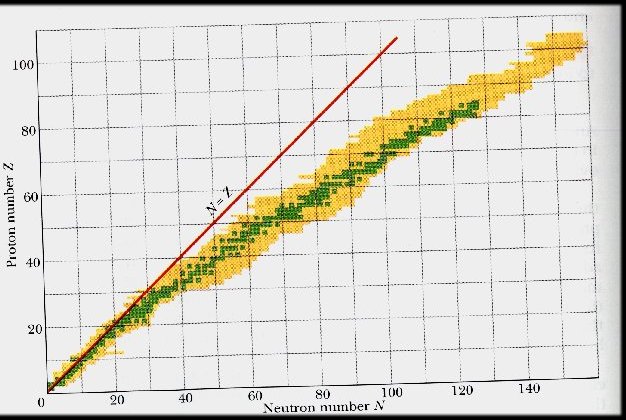
Fig. 10-5. A schematic representation of the periodic table of the nuclei, with the "valley of stability"
Beta Decay: Transmutation of a Neutron to a Proton
If a nucleus has too many neutrons, its protons can be far enough away
from each other that the short-range, strong force binding them together
is overcome by the electrical repulsion of the + charges and the nucleus
can blow apart. Alternatively, an excess neutron can convert to a proton
by emitting an energetic electron (and a neutrino, ?, Fermi's "little neutral
one" that hardly ever interacts, as we will discuss in the next chapter):
n -> p e ?
Note that electrical charge is conserved,
0 = +1 -1 +0
as is momentum and energy, since the neutron is heavier than the proton
by 1.3 MeV, and the electron weighs only 0.5 MeV. The excess energy
ejects the electron and the neutrino at high speeds.
On the other hand, a nucleus with too few neutrons, can simply converts one of the its protons to a neutron, shooting out a positive beta particle (a positive electron, or positron) in the process. Either way, the beta decay nucleus is transmuted to a different element: Nature's alchemy truly at work. Since the force that mediates beta decay is so weak and the lifetimes associated with it so long, it is called the weak force. We will look at it in detail in the next chapter.
Ultimately, heavy radioisotopes decay until they reach the superstability of the iron nucleus, where the nucleons are the most tightly bound of all nuclei. A binding energy of 8.5 MeV is required to rip one out, as is displayed in Fig. 10-6. It is energetically favorable for the heaviest nuclei to decay or fission towards iron, generally an exothermic reaction. On the other hand, nuclei less massive than iron require energy to fuse them together to eventually make iron. (As we will soon learn, the energy to produce the first H and He nuclei came from the Big Bang, the energy to fuse them to make all the elements up to Fe comes from suns like our own, and the additional energy required to jam nuclei together to make transferric elements comes in the shockwave collisions of supernova, exploding stars.)
Fig. 10-6. The binding energy per nucleon as a function of the atomic mass number (amu), showing selected nuclei
Let us lastly consider the gamma ray, the third, and most penetrating of the particles the Curies observed from radioactive decay. In may ways this radiation is the simplest to understand. Remember that electrically charged electrons in an atom deexcite by emitting a photon of light when they rearrange themselves from one wave function to a lower energy wavefunction. Similarly with an electrically charged proton changing from a higher to a lower energy shell in the nucleus. It shakes off a photon of light, only here the difference in energy states is measured in MeV, not eV, so the radiation is a million times more energtic.
The nucleus is a mysterious object, with all three forces of nature active: the strong, electromagnetic and weak. In the next chapter will will explore how these three seemingly different forces may well be different manifestations of the same unified force.
Bibliography
The Particle Data Group of Lawrence Berkeley National Laboratory, Michael
Barnett, Project Leader, The Particle Adventure, http://particleadventure.org.
This provided the inspiration for the text and the source of the graphics
of this chapter.
Chapter 11. The Unification of all Particles and Forces of Nature
To the non-specialist, the physics of the late 20th-century seems to rain with particles: quarks, leptons...fermions, bosons...photons, W's, gluons, gravitons. Ever higher energy collisions created by powerful accelerators give birth to still more massive and bizarre fragments of the stuff which makes up our universe. Meanwhile, sensitive detectors reveal that the heavens themselves continually shower us with cosmic rays containing muons and neutrinos. Can we make sense of this profusion? Is there a logical order? Or is this a chaotic downpour? Nowadays we believe we have a "Standard Model" which accommodates all the experimental findings, has a place for each particle, and expects no new particles. If so, we are justified in "singing in the rain!" And we are of the same mind as the scientists at the close of the 19th-century who claimed that the essence of all science had already been discovered.
In this chapter we explore the concepts central to The Standard Model of particle physics, and how they lead to the vision of the unification of three of the four forces of nature...electromagnetic, weak, and strong, ...and of all the particles they act upon... into one primordial particle. This vision unites the super big and the ultra small, as represented by Glashow's classic snake shown in Fig. 11-1. Glimpsing the ultra small requires the resolution of the highest possible energies, which were only present at the first instants of the hot big bang, when the universe itself was infinitesimally small. Conversely, the super big...the farthest reaches of the universe today... let us glimpse back to the earliest epoch of our space-time, when all particles were one, and all forces the same.
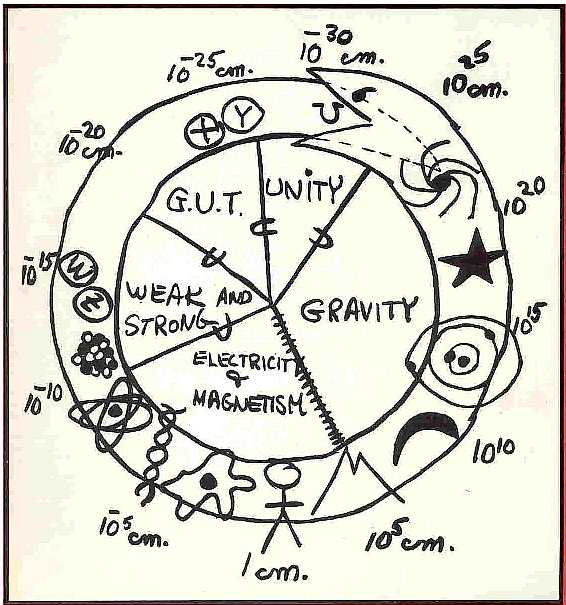
Fig. 11-1. Glashow's Snake: The unity of the forces on the inside and the characteristic structure sizes on the snake, with the succession of scale size on the outside. The snake devours its tail, where the physics of very smallest (the Planck scale, 10-30 cm) is visible by peering back in time to the outer reaches of the universe (1025 cm).
Fermions - the Matter Particles - and the Pauli Exclusion Principle
As we saw in Chapter 7, the periodicity of Mendeleev Table of the Elements (Table 7-1) ultimately revealed that each atom was a composite of a number of smaller entities, electrons. The complex structure of each atom arose because each electron possesses an intrinsic spin of 1/2 (in units of Planck's constant h/2?), behaving like an infinitely small quantum mechanical top. All such identical matter particles, called fermions, obey the Pauli principle: no two identical particles can occupy the same quantum state at the same place and the same time. Hence, as the levels of an atom fill up by adding successively more electrons, the atomic structure of each is different, changing the chemical properties of each new element.
The electron is the paradigm fermion. All matter particles... electrons, neutrinos, nucleons...leptons and quarks... have intrinsic spin = 1/2. Each are subject to the Pauli Exclusion Principle; each can have only one representative in a given quantum state
Bosons as Force-carrying Particles
The electromagnetic force between the mass particles of the atom, the electron and the positively charged nucleus to which it is bound, is mediated by a very different kind of particle, the photon. It is the prototypical force-carrying particle or boson, as we observed with a de-exciting atom; the electron shook off a photon at one atom, the photon traveled to another, and was absorbed by another electron there, promoting it to a higher quantum level.
In contrast with the fermions (the spin 1/2, matter particles), all bosons...photons, gluons, W's and Z's, and gravitons...have integer intrinsic spin, either 0, 1, 2, and are explicitly not subject to Pauli exclusion. Indeed, as many as possible can occupy any one quantum state. For the photon, this leads to the laser, where an initial photon stimulates the emission of as many photons as it encounters to jam into the same direction at the same time with the same frequency.
In the last Chapter we saw a repeat of earlier experience with the atom: the Periodic Chart of the Nuclei, the roughly proportional relationship between the number of proton in the nucleus and its mass (Fig. 10-5), revealed that the nucleus is constructed of fundamental building blocks. Only in 1932 was the second building block, the neutron, discovered by Chadwick.. Again the quantum shells were filled sequentially by adding separately the fermion subparticles, protons and neutrons, yielding different properties for each nucleus.
Are the Proton and Neutron Fundamental?..Strangeness, Charm, and
all that
As soon as the neutron was discovered the question naturally arose: Are protons and neutrons divisible? But they themselves formed a small "periodic chart", a doublet (n,p) now generically called the nucleon. They look almost identical, only differing electric charge, 0 and + They differ in mass by only 0.1%. As anticipated, within two decades excited states of the nucleon and new periodic charts emerged, the Tables of Nucleons, Baryons, Hyperons, and Mesons. The nucleons had a myrad of excitations, analogous to the higher levels of atoms and of nuclei. But these were categorized by a strange new conserved observable. This observable came in integral units, just like electric charge, and behaved very much like a generalized electric charge. For want of a better term, strange charge, or just "strangeness" took scientists' naming fancy.
The allowed particle states formed geometric patterns...triangles
and hexagons...plotted in the two variables, charge and strangeness (horizontal
and vertical axes, respectively, in Fig. 11-2). Three of the many
Periodic Tables are shown in the figure. Notice the (n,p) doublet
in the top of the right hexagon. Like Mendeleev before them, the
scientists of the day were mystified by the "missing" particle, with strangeness
-3, at the apex of the bottom triangle. Dubbed the omega minus, its
discovery cliched the two-dimensional periodicity of the elementary particles,
and gave credence to the new "quark model". Considered only a mathematical
fiction at the time, this equilateral triangle of three fundamental particles
reproduced all the observed states, and none of the unseen states.
But it had what was considered a fatal flaw: the electric charges
of the quarks came in none integral values, -1/3 for the down quark, and
+2/3 for the up. 1/3 charges were unthinkable, and had never been
seen. But eventually they were shown to be true!
Fig. 11-2. Left: Periodic Tables of Mesons (upper), Nucleons (right),
and Hyperons (lower) - circa 1964. Right: The same periodic charts
in 1974, with the addition of the quark tetrahedron at center right.
Charm and the Triumphant Quark Model
The physicists of the day had only to search for a decade more to uncover another totally unexpected quantum number. This, a third generalized electric charge, named "charm", transformed all of the 2D charts into 3 dimensional periodic tables of mesons and baryons, with the charm quantum number providing the third axis. This additional degree of freedom was easily assimilated by the quark model. The (d,u) (down,up) quark doublet now had a replication, the (s, c) (strange,charm) doublet, as is represented by the tetrahedron at center right in Fig. 11-2. Again unexpectedly, in the final decade of the 20th century, a third quark doublet, (b,t) (bottom,top), appeared in the even higher-energy detectors. Finally the quark sector was completely observed, with its three doublets: (d,u), (s, c), (b,t).
The successful search for all quarks (the matter particles subject to the strong force) had its counterpart in the search for those matter particles that do not feel the strong force, the leptons. In fact, the muon (?),the heavy brother of the electron (e), had been discovered in the cosmic rays, even before strangeness appeared. Cloud chamber tracks reveal that a muon, borne of particles from the cosmos, pierces each of us at the rate of one every second. Since the muon was completely unanticipated, its appearance prompted Pauli to complain, "Who ordered that?" A third heavy lepton, the tauon????, was observed soon after charm (its mass is similar).
Each of the three electrically charged leptons has an associated neutral partner, a neutrino (?), which always accompanies it in weak (beta decay) reactions. These partnerships are codified as three more conserved, generalized charges. By convention, the normal leptons in nature, e.g. the electron and the electron-neutrino (?e) are assigned an electron number of +1. Conversely, the electron number of the positron and antielectron-neutrino is -1. The muon and tauon numbers of all four are 0. In summary, the lepton sector, like the quark sector, has three matter doublets: (e,?e), (????), (?????).
But will it stop here? Yes, we think so. No new periodicity has been seen. Evidence from the cosmos, which we will discuss in the next chapter, limits the total number of particle generations (doublets) to three. And, almost a century later, thanks to a new generation of Rutherford scattering experiments, we observe no evidence of compositeness in any of the quarks. By slicing the nucleon into 10,000 pieces, we find no indication of substructure in the quarks, as well as the leptons. All are pointlike to an incredible degree of accuracy, using the highest resolution of the best microscopes, our high energy accelerators.
The Standard Model and The Periodic Chart of Fermions
Thus we arrive at The Standard Model, which describes all experiments on matter particles by non-gravity forces. It quantitatively explains hundreds of complex particle states and their complex interactions with incredible accuracy. The fundamental entities of the Model are the following:
Matter Particles (in three generations of doublets) = spin 1/2
fermions:
leptons for non-strong interactions (em and weak)
quarks for strong interactions (as well as em and weak)
Force Carrier Particles (one/fundamental interaction) = integer
spin bosons:
electromagnetic: a singlet photon(?)
weak:
a triplet W+-/Z°
strong: an octet
gluon (g)
The "Periodic Chart of Fundamental Particles" can be configured
in many ways, with one of the most elegant for the matter particles displayed
in Fig. 11-3. Its organization reflects the current picture of the
unification of particles, where all these particles derive from a single
primordial one at the earliest times in the universe, when the highest
energies and temperatures prevailed. The doublets are arrayed vertically.
Similar properties e.g. electric charge, occur in rows. Mass
increases to the right, with each column representing a generation.
Hence, decays of heavy particles into lighter ones proceed from the right
to the left.
Fig. 11-3. The fundamental matter particles of The Standard Model.
Less unified earlier Tables of the Quarks, Leptons, and Bosons appear in Fig. 11-4, where the properties of all of the known particles are explicitly recorded, including those of the bosons. Note the absence of the yet undiscovered graviton at the right of the Boson Table. It is the putative carrier of the gravitational force. A new generation of gravity wave antennas is currently under construction, so the graviton should take its rightful place in the table within the decade.
The Weak Interaction and Our Sun
If nature gives us 6 kinds of quarks and 6 types of leptons, why do we only see evidence of the up and down quark (the proton and neutron) and the electron? Thanks to the weak interaction(via its W and Z force-carrying bosons), more massive quarks and leptons decay into lighter ones. And lucky we are. The weak force powers the energy producing sun. And, just as important, the weakness of radioactive force is responsible for its longevity. Without the slow, weak interaction to preserve a constant sun for 4.5 billion years, we would not have had sufficient time to evolve as a species
Where does the sun get its power? Nuclear fusion, converting mass to energy, is the ultimate source. The fusion reaction of two protons (p) in the sun to form a deuteron (d) (the nucleus of heavy hydrogen, the bound state of a proton and a neutron) goes as follows:
p + p -> p n e+ ?e -> d e+ ?e
The presence of the telltale neutrino along with its partner positron explicitly reveals the action of the weak force.
Radioactive Beta Decay, the W Boson, and Virtual Particles
In the last chapter we encountered the inverse of this reaction, the
radioactive beta decay of the neutron. In that classic weak interaction,
a d quark in a neutron (udd) decays to a u quark making a proton (uud).
The "beta-ray" electron (to conserve electric charge) and an electron-antineutrino
(to conserve electron number) are emitted in the process. A graphic
description of beta decay, highlighting the action of the force-carrying
intermediate W boson, is drawn in Fig. 11-5. This process was utterly
mystical until two decades ago when the W boson was discovered. The
critical role of the W is hidden from us since its mass is 100 times that
of the decaying neutron. Hence it has only a fleeting existence and
is invisible to the scientist.
Fig. 11-5. Graphic representation of the weak beta decay of a
neutron. All the particles are pointlike, so ignore the size imputed
to them by the artist.
The process in Fig. 11-6 is commonly drawn by physicists as a Feynman
graph, which represents a mathematical integration that calculates the
probability amplitude for the transition to occur. Fig. 11-7 displays
the Feynman graph for a similar reaction, a d quark transforming into a
u quark, and radiating a W- in the process. The virtual W- travels
a short distance, consistent with the Heisenberg Uncertainty Principle,
and decays into a ?? and an anti-???
Fig. 11-6. The Feynman diagram representing the radiation of a
W boson and its subsequent leptonic decay into muon and muon anti-neutrino.
How can a neutron decay into a proton by way of such a massive, "virtual" boson? Isn't conservation of energy violated? Yes, but the W exists so briefly that no "rule" broken. As we learned from Heisenberg's formulation of quantum mechanics, it is impossible to measure both time and energy/mass exactly. Hence, for a brief enough instant, consistent with the uncertainty principle, a particle of any mass can exist.
The Neutrino; Antimatter
A paragraph about the elusive neutrino is in order. This companion of the charged lepton in weak interactions has no charge, and only a minuscule mass, which has just been discovered. Neutrinos almost never interact, passing right through earth without interacting. Just as with their partners the muons, one every sec from the cosmic rays passes through your body, yet in your lifetime only one will ever interact inside you . The presence of the neutrino is evident in weak reactions by its missing momentum, e.g. in beta decay the sum of momenta of electron and proton not equal to the original momentum of the neutron. The decay looks like a Mercedes star, missing one spike. The neutrino carries away the missing momentum. Weakly interacting as they are, neutrinos are critical to our existence. For example, 99% of the energy produced in stars, and also by supernova explosions at the end of a star's life, is emitted in the form of neutrinos.
It is also appropriate to discuss antimatter a bit more. For each matter particle in the quark/lepton table of Fig. 11-3 there is a corresponding antiparticle. An antiparticle is the same as its corresponding matter particle except that all its generalized charges are opposite, e.g. the anti-electron (e+), a positron, is electrically positive; the electron (e-) is electrically negative. Since the electron carries electron number +1 relative to weak interactions, the positron is assigned -1 electron number. However, both have exactly same mass and are affected in the same way by gravity. Normally scientists denote an antiparticle by drawing a bar over its symbol.
When particle and antiparticle meet, annihilation into pure energy occurs. Conversely, pure energy may materialize into any other particle/anti-particle pair or alternatively into a neutral force-carrier particles, such as two photons, a virtual Z boson, or a (colorless) gluon state. If the reaction is energetically favorable, a particle / antiparticle pair materializes out of the vacuum.
Annihilation of positrons by electrons in your body has yielded a powerful technique to tomographically locate minute tumors before they metastasize. Many back-to-back annihilation gamma rays are reconstructed to the region where radioactive 22Na salt (the source of the positrons) is being rapidly metabolized by the tumor.
The Strong Interaction and its Gluon Carriers
Having explored the electromagnetic and weak interactions, we now turn to the strong interaction. The carriers of the three forces, the ?, the W/Z, and the gluon g, are all spin 1 bosons. Further, they are representations of force in three different mathematical dimensions. For the em and weak forces,
?
(W- Z° W+)
the photon is a singlet and the W/Z a triplet. By a continuation that is suggestive of the unification of the three forces, the gluon is a 3 x 3 matrix, a nonet. (Technically, one constraint exists on the nine original gluon states, so actually only 8 independent gluon states exist.) The only particles that participate in the strong force, the quarks and the gluons, exchange a generalized charge that is the source of the strong force. This interaction is described by the same mathematics that explain visual color. Hence physicists have dubbed this generalized charge the "color" charge, even though it has nothing to do with the colors of light.
Each gluon carries two values of the strong charge, color and
anti-color. Since all colors can be represented by the three primary
colors (red, green, and blue), nine possible combinations of color and
anti-color exist, as displayed in Fig. 11-7. In the figure we distinguish
anti-color from color by representing the anti- in italics.
Fig. 11-7. The 9 coloranti-color states of the gluon.
The force mediated by color-charged gluons is very strong, e.g. holding the average of three quarks together in the form of a proton or a neutron. Only quarks and gluons carry color charge. All strongly interacting particles that live for more than a fleeting existence, e.g. the proton and neutron, are color neutral, or "white" objects. Hence, on average, one each of the three quarks in a proton must carry red, green, and blue color charge. And in the emission of a gluon, conservation of color charge means that two of the three quarks change color. Only the gluons along the diagonal of the matrix above are color neutral, "white" bosons, so the exchange is usually one that is "colorful" and must occur rapidly enough that it does not violate the Heisenberg principle. (All fermions that do not feel the strong force, i.e., the leptons, are also "white".) Despite its prodigeous strength, the strong force only acts at very small distances. At lengths bigger than the uranium nucleus (the biggest stable nucleus) the gluon exchange no longer overpowers the electromagnetic repulsion between two protons.
Unification of the Forces
Scientists yearn to simplify their models of the universe. They are driven to find unifying principles. The first step in recognizing that all forces may have one primordial precursor was taken by Newton, who unified planetary and terrestrial gravity. The same law explained an apple falling to the ground and the moon perpetually falling toward the earth. The second unification of forces, fusing electricity and magnetism, was accomplished by Maxwell. He recognized that electric charge (+/-) and magnetism (north/south poles) are different aspects of the same force. The third step, the Unified Electroweak Interaction, occurred a quarter century ago. Weak charges are closely related to electromagnetic charge. For example, the Z° of the weak force and the ? of the electromagnetic quantum mechanically interfere. At very short distances, the weak interaction becomes comparable in strength to the electromagnetic. The apparent difference in strength in our current, cold world is due to the artificial, though huge difference in mass between the W/Z (100 times that of the proton) and massless photon. However in a super hot world, where temperatures exceed the mass of the W/Z, the distinction between the two forces vanishes.
Work on the final two unifications is well underway. Evidence for the first, the union of the electromagnetic, the weak, and the strong force into a Grand Unified Theory (GUT), is shown in Fig. 11-8. Measurements of the strengths of the three forces reveal that they begin to merging even at very low energies (the far left). The GUT theory extrapolates these forces to high energies: all three forces converge to one strength at a unique high energy (or at an early time in the history of the universe). All three interactions appear to be different aspects of the same unified interaction.
According to Heisenberg, the primordial force-carrier particle,
massive though it may be (1016 GeV on the graph), could exist for the briefest
of instants. It would cause quarks to transform into leptons (inducing
transitions between the states in Fig. 11-3). The result: the
proton would decay into a positron and two quarks. Examples of this
reaction are being sought with the world's largest detector, 50,000 tons
of ultra pure water a mile underground. This reaction had to
have occurred sometime in the history of the universe, and must have occurred
faster for antiprotons than for protons. Otherwise our matter-rich
world (we see no antimatter around us) would not exist today. Our
universe is now dominated by photons, a billion times more of them than
protons. But we observe essentially no antiprotons. Clearly
protons and antiprotons both annihilated each other and decayed, with the
antiprotons decaying faster than the protons. A major problem for
unification to solve: Why did matter "win" over antimatter, when
they are normally exactly equal and opposite?
Fig. 11-8. The evolution of the strengths of the strong, weak
and em forces with energy.
Another major unsolved problem beyond the Standard Model is that of mass. What is its origin? Why do the particles have such wildly different masses, as depicted in Fig. 11-9? Why, for example, is the t quark 200 times heavier than the proton, and 400,000 times more massive that the electron? The electroweak unification theory suggests that a new boson, the Higgs boson, is responsible for mass. The world's largest atom smasher, the LHC in Geneva, is being constructed to discover it.
Since mass is the generalized charge that is acted upon by gravity
(the graviton), solving the problem of mass entails solving the unification
with gravity. The mathematics for the last unification, the union
of the GUT force with gravity, has been a hotbed of activity for the last
decade. It is called string theory. This theory envisions a
universe where every point in spacetime contains little loops, dimensions
that did not expand like spacetime when the universe expanded. Instead,
these loops encode at that point in spacetime all the intrinsic properties
of the particles there...their mass and quantum numbers.
Fig. 11-9. Graphical representation of the mysterious mass spectrum of the fundamental particles. The area of the circle for each particle is proportional to its mass. Caution: the scale for the bosons at the left is in GeV; the scale for the fermions on the right is in MeV.
These two unifications, the Grand Unified Theory and the string gravitation theory, as well as the discovery of the last anticipated particles, the Higgs boson and the graviton, are the four Nobel prizes awaiting the scientists in your generation.