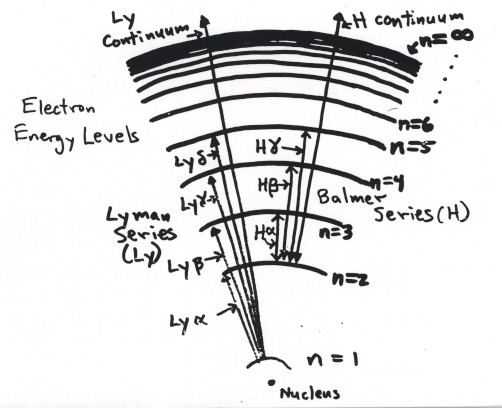
Bohr Hydrogen Atom (1913)
(not to scale!)

n = 1 lowest energy level; higher levels more
closely spaced
each energy level specific electron energy
bright line emission of photon:
electron jumps down, from upper to lower unoccupied level
e.g. n = 3 to n = 2, the Hydrogen a famous red line
Absorption (dark) line:
photon kicks electron up from lower to higher unoccupied level photon is annihilated